- Business
- Technology
Latest in Technology
Israeli Study Pinpoints Lunar Ice Sites to Guide Future Moon Landings
2 hours ago
The IDF attacked another petrochemical complex in Shiraz – one of the few remaining complexes for the production of essential chemical components for explosives and materials for ballistic missiles.
6 hours ago
We saw that a new battalion was established this month, so we went out on a combat reconnaissance mission.
22 hours ago
Israeli Startup Q-Factor Raises $24M to Build Million-Qubit Quantum Computer
23 hours ago
- Politics
Latest in Politics
We will continue to deepen the damage to the Iranian regime and stabilize a forward defense area in Lebanon to prevent direct fire at our communities.
3 hours ago
The IDF eliminated the chairman of the Revolutionary Guards’ intelligence organization in Tehran. The Air Force, with precise intelligence guidance from Aman,
1 day ago
Another blow to the military buildup of the Revolutionary Guards: The IDF eliminated the head of commerce in the Revolutionary Guards’ oil headquarters in the Tehran area, with the Air Force acting on intelligence guidance.
2 days ago
In a precise strike in Tehran: The IDF eliminated the commander of the Iranian terrorist regime’s oil headquarters. The Air Force, with precise intelligence guidance from Aman,
5 days ago
- Security
- Economy
Latest in Economy
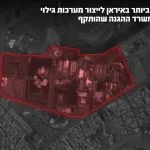
Following the elimination of senior figures and attacks on airports: The IDF attacked Iran’s largest petrochemical complex. The Air Force, under intelligence guidance from Aman,
1 day ago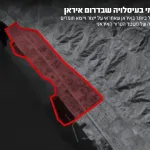
Tel Aviv Stock Exchange TASE Recap April 6, 2026
1 day ago
Israel Restarts Leviathan Gas Field After Five-Week Shutdown
1 day ago
$31 Million in Extra Funding Allocated to Aid Local Governments Dealing with Ongoing War
7 days ago
- Crime
Latest in Crime
Ramallah journalist arrested by Israel Police; probe ongoing
5 hours ago
Two Military Officers and One Civilian Indicted on Charges of Smuggling Prohibited Goods Into Gaza
7 hours ago
Network Aiding Palestinians Enter Israel Illegally Uncovered, Leader Arrested
10 hours ago
Suspect arrested for damage, house arrest violation
10 hours ago
- Society
- Services
Travel & Transportation
Technology & Utilities
Religious & Cultural
Government & Public Services
Information & Directories
Photo by Kobi Richter/TPS-IL on 26 October, 2017
By Pesach Benson • 8 April, 2025
Jerusalem, 8 April, 2025 (TPS-IL) — Israeli researchers revealed that the way bacteria move plays a direct role in the spread of antibiotic resistance on Tuesday in a study that opens up new possibilities for fighting antibiotic resistance.
The research centered on bacterial conjugation, the process by which bacteria share genetic material, including antibiotic resistance. Hebrew University scientists led by Professors Sigal Ben-Yehuda and Ilan Rosenshine found that the rotation of the bacteria’s tail-like flagella acts as a mechanical signal triggering the conjugation process.
Till now, the prevailing understanding was that bacterial conjugation only took place on solid surfaces and that the tail’s rotation was only for movement.
But the findings, recently published in The EMBO Journal, a peer-reviewed journal, found that the tail’s movement, called flagellar rotation, takes place in liquid environments. The researchers also discovered that the tail’s rotational motion acts as a mechano-sensing mechanism causing donor cells to form multicellular clusters with recipient cells to facilitate efficient gene transfer. This helps the bacteria spread resistance genes, even in conditions previously thought to be less favorable for conjugation.
“This process of DNA transfer, called bacterial conjugation, has long been studied on solid surfaces,” said Prof. Ben-Yehuda. “What we found is that in liquid, it’s the rotation of the flagella that acts as a mechanical signal to kickstart this process.”
The study focused on pLS20, a widely distributed plasmid found in Bacilli subtilis, a common soil bacterium.
Researchers observed that rotating flagella triggered gene expression in donor cells, prompting them to form clusters with recipient bacteria. These clusters bring cells into close contact and facilitate DNA transfer. When flagellar rotation was blocked — either through genetic modification or by increasing the viscosity of the surrounding liquid — conjugation rates dropped sharply.
“It’s not just about having flagella,” Ben-Yehuda explained. “They need to rotate. That mechanical action is essential for signaling the bacteria to start sharing DNA.”
Since flagellar rotation is essential for triggering gene transfer, it presents a new potential target for antimicrobial strategies. Disrupting this mechanical signaling could help prevent the spread of resistance genes without killing bacteria outright.
The understanding that bacterial behavior in liquid environments such as the bloodstream, lungs or water systems is more dynamic than previously thought could also lead to better predictive models of how antibiotic resistance spreads.
“Our study brings about a novel notion that synchronizing DNA transfer with the bacterial motile lifestyle provides the plasmid with the advantage of spreading into remote ecological niches,” said Ben-Yehuda.